MEMBRANE
At the cutting edge of technology and innovation, collagen membranes have a triple role to play in GBR (Guided Bone Regeneration): mechanical, physical barrier and preventive.
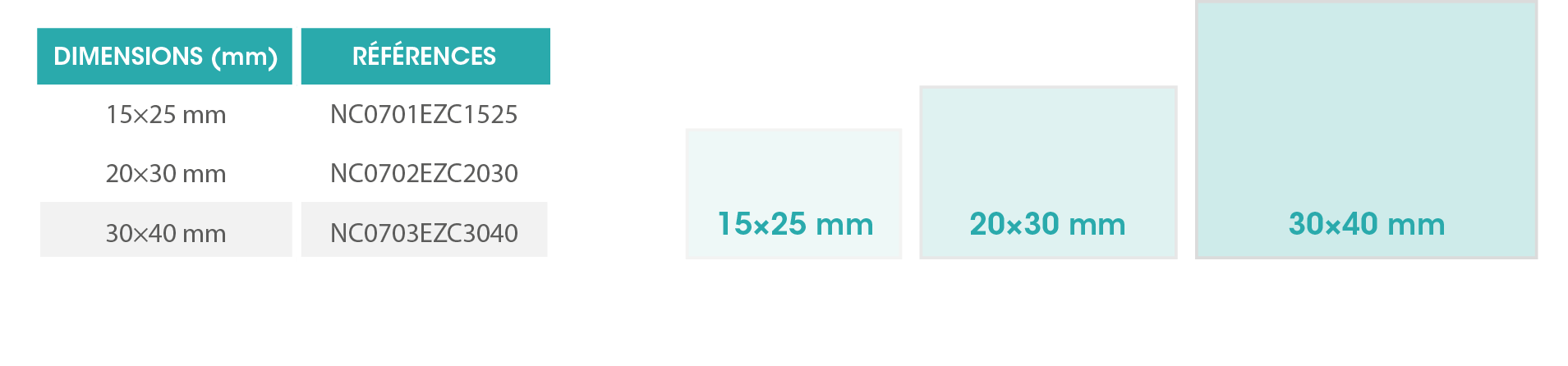
NEA COVATM
AN ABSORBABLE HEALING MEMBRANE
Nea Cova™ is a healing and resorbable membrane made of highly purified porcine collagen (type I and III). It is designed for Guided Tissue Regeneration (GTR) and Guided Bone Regeneration (GBR) procedures in dental surgery. Phylogenetically, porcine collagen is the closest to human collagen. Porcine tissue is considered as a material of excellence in many medical xenograft procedures.

The Nea Cova™ resorbable membrane offers excellent handling property. Its adaptability to different bone geometries enables easy and effective surgical procedures.
Barrier function for 3 months
Easy to cut, handle, position & readjust
Excellent adhesion
Resistant, suturable and sandy pine
Flexible & adaptable to different bone geometries
Resorbable

CONSULT THE DOCUMENTATION
Discover all benefits of the Nea Cova membrane.
Manufacturer: Biomatlante SAS.
Distributed by: Biotech Dental. BIOTECH DENTAL – 305, Allées de Craponne – 13300 Salon de Provence – France. simplified company limited by shares with a capital of €24,866,417 – trade and companies register of Salon de Provence, France: 795 001 304 – SIRET number [French business registration number]: 795 001 304 00018 – VAT No.: Implantable sterile medical devices marked CE31 Class III for surgery; reserved for health professionals.
Read the instructions in the package leaflet and on the label carefully before use. Images for illustration purposes only. These medical devices are regulated health products that carry the CE mark.